On this page
-
Text (2)
-
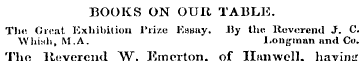
Untitled Article
-
Untitled Article
Note: This text has been automatically extracted via Optical Character Recognition (OCR) software. The text has not been manually corrected and should not be relied on to be an accurate representation of the item.
-
-
Transcript
-
Note: This text has been automatically extracted via Optical Character Recognition (OCR) software. The text has not been manually corrected and should not be relied on to be an accurate representation of the item.
Additionally, when viewing full transcripts, extracted text may not be in the same order as the original document.
Untitled Article
fcew products / ' If this be bo , what is there unreasonable in the supposition that trituration may develope new forces in the medicines ? Our stupendous ignorance of chemical action—especially of organic chemistry—renders it imperative on us to be cautious ere we front a fact with any denial derived from our philosophy . The homoeopathist proclaims his fact ; examine if it be true , if the fact be bo ; but do not reject it because monstrous in academic eyes . Our readers know that we have ( riven no pledge to homoeopathy : we only desire
tihat it may be treated with the same respect as other tentatives to get at a solution of the mystery . Our purpose in commenting on Liebig ' s assault was , however , ulterior in its aim . "We wished to exemplify how , from the want of real philosophy , a man could sneer at an opinion which his scientific experience tended to support . In Chemistry he was familiar enough with the importance attributable to mere change of position ; but in Medicine he stuck to the old routine , and laughed at the idea of trituration and dilution producing any
new effects ! But enough of controversy ! Liebig ' s work is not controversial , and we but ill convey an idea of it by touching on these points . It is substantially what the title imports—a Familiar Exposition of Chemistry , setting forth with very great clearness all the leading principles accepted by men of science , and illustrating them with abundance of detail , much of it very curious , and drawn from the most recent discoveries . The book is too compact to admit of analysis in any space that we could afford it ; but to the passages already quoted we shall add two , as samples of his exposition : —
KOSMOS . " Until very recently it was supposed that the physical qualities of bodies , —i . e ., hardness , colour , density , transparency , &c , must depend upon the nature of their elements or upon their composition . No one could imagine to himself one and the same body in two different states , and it was tacitly received as a princip le that two bodies containing the same elements in the same proportion must of necessity possess the same properties . How could it be possible , otherwise , for the most ingenious
philosophers to regard chemical combination as an interpenetration of the panicles of different kinds of matter , and matter aB susceptible of infinite division ? There never was a greater error . If matter were infinitely divisible in this sense , its particles must be imponderable , and billions of such molecules could not weigh more than an infinitely email one . But even the particles of that imponderable matter , which , striking upon the retina , produce that sensation which , when it has reached the inner consciousness , is recognized as light , are not , in a mathematical cense , infinitely small .
" Inter-penetration of elements in the production of a chemical compound supposes two distinct bodies , A and B , to occupy one and the same upace at the same time . If this were so , different properties could not consist with identity of composition . ' That hypothesis , however , has shared the fate of all the -views of natural phenomena entertained by the philosophers of past times . It him fallen , like them , without any one taking the trouble to maintain it . The force of truth , dependent upon observation , is irresistible . A great many substances have been discovered amongst organic bodies , composed of thu
same elements in the same relative proportions , and yet exhibiting physical and chemical properties perfectly distinct one from another . To such substances the term Isomeric ( from lcr& 4 equal , and pepo ; part ) is applied . A great class of bodies , known as the volatile oils—oil of turpentine , essence of lemonp , oil of bulsum of copaiba , oil of rosemary , oil of juniper , und many others , differing widely from each other in their odour , in their medicinal effects , in their boiling point , in their specific gravity , &c , contain the Hume elements , carbon and hydrogen , in the same proportions . No one of them contains more of either element tluin the otlierH do .
? ' llow admirably aim pie does the chemkrtry ol organic nuturo present itself to uh from this point of view ! An extraordinary variety of the ino . st remarkable compound bodies is produced with equal weights of two elements—and how wide their din-Bimliarity ! The crystallized part of the oil of rosi'H , tin * delicious fragrance of which i « ho well known , a solid at ordinary temperatures , although readily volatile , is a compound body , containing exactly the
Aitme elements and in the same proportion * an the » h we employ for lighting our streets ; and , in short , the unto elements , in the unmo relutive quantities , are found 4 H * defon Other compounds , all differing chm : ii-Ts . lbLiB ^ in tn . OijrTphy Hical uud (; ht ; micul properties - " ThC * e ^ emarkal * le truths , no highly important in ' tlioir app lication * , wfcre not received and admitted an / sufficiently eatuVlwhM , without hufhcient proofs . I M » pt s ample ' s havd long been known where the \; 6 * iatyB } B of two Aiffbtlant bodies gave the « ame com' position ; but suck okneB were isolated observations ,
homeless in the realms of science ; until , at length , examples were discovered of two or more bodies whose absolute identity of composition , with totally distinct properties , could be demonstrated in a more obvious and conclusive manner than by mere analysis ; that is , they can be converted and reconverted into each other without addition and without
sub-. " In cyanuric acid , hydrated cyanic acid , and cyameHde , we have three such isomeric compounds . " Cyanm-ic acid is crystalline , soluble in water , and capable of forming « alts with metallic oxides . " Hydrated cyanic acid is a volatile and highly corrosive fluid , which cannot be brought into contact with water without being instantaneously decomposed . " Cyamelide is a white substance very like porcelain , absolutely insoluble in water . acidin
" Now , if we place the first , cyanuric , a vessel hermetically sealed , and apply a high degree of heat , it is converted by that influence into hydrated cyanic acid ; and then , if this is kept for some time at the common temperature , it passes into cyamelide , no constituent being separated , nor any body taken up from without . And again , inversely , cyamelide can fee converted into cyanuric acid and hy drated cyanic acid . *« We have three other bodies which pass through similar changes , in aldehyde , metaldehyde , and elaldehyde ; and again two , in urea and cyanate of ammonia . Further , 100 parts of aldehyde , hydrated butyric acid , and acetic ether , contain the same elements in the same proportion . Thus one substance may be converted into another without the separation of anv of its elements , and without the introduction
of any foreign body . " The doctrine that matter is not infinitely divisible , but , on the contrary , consists of atoms incapable of further division , alone furnishes us with a satisfactory explanation of these phenomena . In chemical combinations , the ultimate atoms of bodies do not penetrate each other , they are only arranged side by side in a certain order , and the properties of the compound depend entirely upon this order . If they are made to change their place—their mode of arrangement—by an impulse from without , they
combine again in a different manner , and another compound is formed with totally different properties . We may suppose that one atom combines with one atom of another element to form a compound Etom , while in other bodies two and two , four and four , eight and eight arc united ; so that in all such compounds the amount per cent , of the elements is absolutely equal ; and yet their physical and chemical properties must be totally different , the constitution of each atom being peculiar , in one body consisting of two , in another of four , in a third of eight , and in a fourth of sixteen ample atoms . "
l'HYSIOLOGY AND CHHM 1 STKY . " Through Nature herself , who is a whole , the natural sciences stand in a necessary mutual connection , so that no one of them can entirely dispense with all the others for its development . The extension of the individual branches of science by researches , has the inevitable result , that in a certain stage , or at a certain period , two of them , for example , come into contact at their boundaries . As a general rule , anew science arises on the debateable land between them , which combines in itself the objects and the modes of viewing the phenomena of both . In order to this interpenotoation , both must have reached a certain
advanced Btage ; the independence ot the original territories must be secured , for till this be done , the energies of the philosopher will not be applied to the border province . In these days we look forward to such a fusion of physiology with chemistry , as to one of the most striking results of Hcientific investigation . Physiology hay attained a point at which it can no longer dispense with chemistry in striving after its object , namely , the study of the vital phenomena in their natural succession . Chemistry , the duty of which is to show in what degree the vital properties depend' on chemical forces , has been prepared , and in now ready , to take up new departments of science , to be independently Btudied . »
" The phenomena presented by animals during their life are among the most complicated natural appearances ; and the detection of their different causes , and the aWertnining the precise share of each in the result , in u tusk of peculiar difli < ulty . "It is a rule in natural science to divide every difficulty which is to be examined , into an many parts an possible , and to study each ol these separately . According to thin rule , all physiological phenomena may be divided into two classes , of which each , up to a certain point , may be studied quite independentl y of the other . Such a . scpiiration , it in obvious , is not found in Nature , where both classes of phenomena are mutually dependent , so thut , indeed , they mutually determine each other .
"The processes of impregnation , development , and growth in animals , the mtitunl relntions of their organs , und the agencies peculiar to these ,- —the laws of their motion , and of that of the fluids of tin , body —the anatomical and other peculiarities of nervous and of musculnr fibres ; all them ? striking and
interesting phenomena may be ascertained without regard to the nature of the substances which form the parts in which these properties reside . But physiology lias to do with other phenomena , not less important . Digestion , sanguification , nutrition , respiration , and secretion depend on a change of form and quality of the substances introduced from without into the system , or on certain solid or liquid constituents of the organism ; and it is in the study of these processes .
as far as they can be regarded apart from structure , that chemistry must come to the aid of physiology . It is evident that physiology has two foundations , and that by the fusion of physiological physics , the foundation of which is anatomy , with physiological chemistry , which rests on animal chemistry , a new science must arise , a true physiology , which will stand in the same relation to the physiology of the present day , as modern chemistry does to that of the eighteenth
century . " In order to form a just idea of the interpenetration of physiology and chemistry , we must call to mind similar occurrences in the history of science . Thus the character of modern chemistry has been essentially determined by the circumstance that it has absorbed into itself entire branches of physics , which now no longer belong to that science . The density of bodies in the gaseous state , forty years ago , was regarded as a purely physical character ; but since we have learned that this property depends on
the composition in a fixed relation , the study of this property belongs to chemistry . Similar relations have been discovered between the specific heat , the boiling point , and the crystalline form of bodies , on the one hand , and their composition on the other ; and it is now chemistry which especially occupies itself with the exact determination of these properties . The doctrine of electricity , so far as it is the result of a change in form and quality , of a chemical change , has now almost entirely passed into the domain of chemistry .
" Exactly in the same way , the more accurate knowledge of vital phenomena will establish the conviction that a number of physiological properties depend on chemical composition ; and physiology , when it shall have takeu up animal chemistry , as a part of itself , will possess the means of investigating this relation of dependence ; it will then be enabled to find a juster expression for physiological
phenomena . Men have often tried to explain vital phenomena exclusively on chemical principles , and to make physiology a part of chemistry . This was done centuries ago , at a time when the chemical changes in the body were better known than the organism itself . But when men had learned to know the admirable structure , the form and quality of the organs , and their combined action by a more exact study of anatomy , they imagined that they had found the key in certain principles of mechanics . All such attempts have entirely failed ; and their failure gave rise to physiology as an independent science .
Mineralogy was in a similar relation to chemistry ; forty years ago , many considered it a part of chemistry , and compound minerals were classed with the salts . Mineralogy conquered her independence , not by rejecting the doctrines of chemistry , but by taking into her own domain the determination of the composisition of minerals . Since mineral analysis has become a part of mineralogy , it is from mineralogists that we have obtained , and daily obtain , the most valuable results in regard to the relation which exists between the chemical composition of minerals and their crystalline form and other physical properties . "
Untitled Article
BOOKS ON OUR TABLE ,. Tin ; Great Exhibition Prize Katmy . JJy the llevorend J . C . Whiuh , M . A . liOiig-inan and Co . The Reverend W . Emerton , of Hanwell , having offered a prize of £ 10 / 5 for the best essay on " The Respect in which the Exhibition wns calculated to further the Moral and Religious Welfare of Mankind and the Glory of God , " it was ultimately awarded to the lteverend Mr . Whish . The performance is a sort of sermon , marked by much feeble good nuture . The author ' s point of view being the pulpit , he delineates chiefly the spiritual aspect of the question ; but had he also taken the workshop and the cottage , as his points of view , the object of the Reverend donor might have been more efficientl y answered . Key to tluj ( Jruat Exhibition , 1 HTjI . Ity 10 . Heine . Ackcrinnnn und ('<> . The plan is clever and interesting , the sheet representing in its lines and colours the various divinionfi of the Crystal J ' nlaee . The avenuos are . represented by a white ground , the compartments by a green , ana the galleries by a pink shading . A list of the chid objects of interest is also printed in each compartment , so that at a glance the visitor may discover hiw way through the building , and to its chief points ol attraction . It will soon appear in several languages-Mjmh Muriiiiuau and her Muutcr . Ity J . 8 tov < -n « on UuBhimi ' . M . I ) . John Churchill-The tone of thin work is so extremel y objectionable—ho unbecoming the gravity of p hilosophic diucusHion and the courtesies of literature—that vvo cannot suffer it to putss without calling Dr . Bushm *
Untitled Article
70 S VL % t ILeaitt * [ Saturday ,
-
-
Citation
-
Leader (1850-1860), July 26, 1851, page 708, in the Nineteenth-Century Serials Edition (2008; 2018) ncse2.kdl.kcl.ac.uk/periodicals/l/issues/vm2-ncseproduct1893/page/16/
-